Protein extraction from RIPA Buffer insoluble fraction
ULTRARIPA kit for Lipid Raft
Highlights
-
Recovering insoluble proteins from RIPA Buffer pellet including Lipid Raft proteins.
-
Little effect on protein structures and functions.
-
Applicable to both of tissues and cells.
-
Easy and Simple Procedure : Only a centrifuge is required.
Product Information
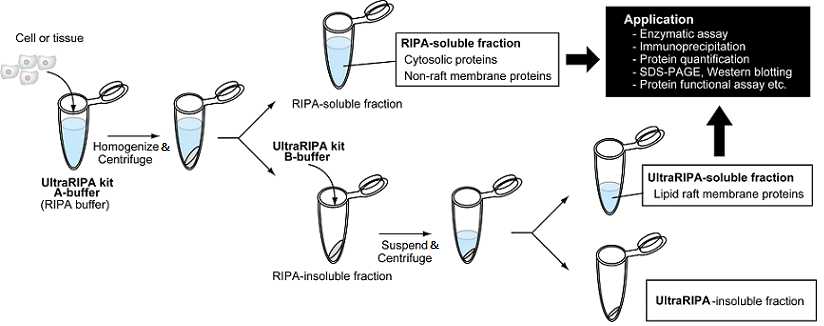
RIPA (Radio-ImmunoPrecipitation Assay) buffer is one of the most useful buffer for protein extraction from cells and tissues. Whereas sodium dodecyl sulphate (SDS)-containing buffer disrupts protein structures, RIPA buffer keeps almost native structures of proteins and can be applied to various applications. However, RIPA buffer is not sufficient to extract membrane proteins and membrane-associated proteins concentrated in lipid raft (Figure 1). Lipid raft is a highly specialized microdomain on the lipid bilayer which contains specialized lipids (sphingomyeline etc), cholesterol and functional proteins. Synapses in neurons, immunosynapses in immunocytes, tight junctions in epithelial cells and focal adhesions in adherent cells are major examples of the lipid raft. These lipid rafts are also called “Detergent resistant membrane (DRM)”, because proteins enriched in lipid raft usually insoluble by mild detergent buffers such as 1% TritonX100 and RIPA buffer. Consequently, it is difficult to analyze functions of lipid raft proteins by RIPA buffer. ULTRARIPA® kit, which consists of two types of detergent containing buffers, can efficiently and rapidly extract membrane proteins/membrane-associated proteins enriched in lipid rafts with native structure and native function. ULTRARIPA® kit is totally new buffer not containing protein denaturing detergents, but can extract the DRM which was hard to extract with RIPA buffer. ULTRARIPA® kit enables to analyze various biological assays of the protein in lipid raft, because enzymatic assay, protein-protein interaction assay etc. is applicable.
Outline of procedure
-
Solubilize samples (tissues / cells) with A Buffer (RIPA Buffer)
-
Centrifuge (>10,000 x g, 5 minutes) and collect insoluble fraction
-
Add B buffer to insoluble fraction
-
Centrifuge (>10,000 x g, 5 minutes) and collect soluble fraction
-
Ready for further assays

<Application 1>
Extraction and functional analysis of the synaptic protein PSD-95 from mouse brain
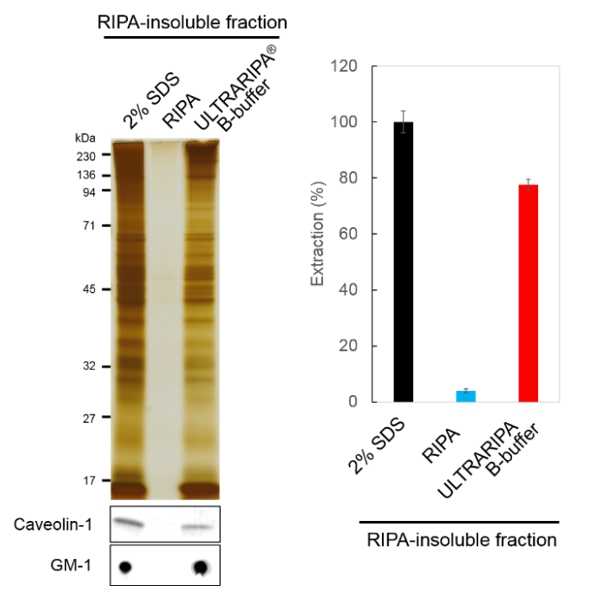
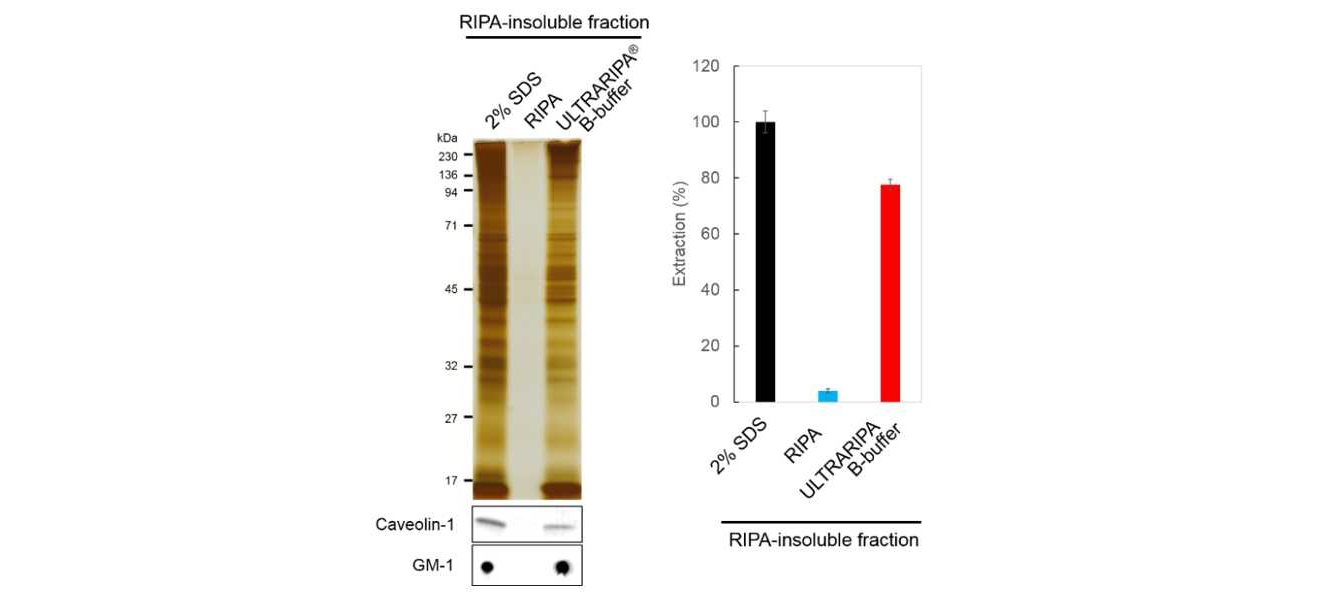
Mouse brain was extracted with RIPA Buffer and insoluble fraction was further etracted. With ULTRARIPA Buffer, >70% extracted proteins from RIPA-insoluble fraction was observed compared to 2% SDS extraction buffer. Lipid Raft markers (Caveolin-1 and GM-1) were also detected.
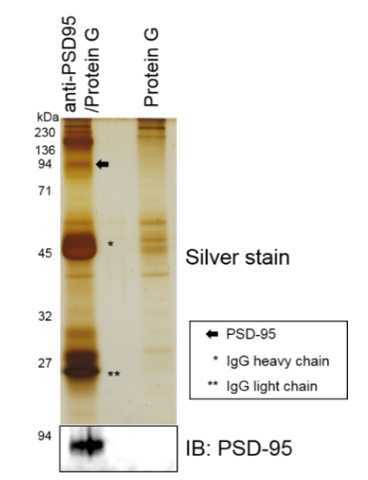
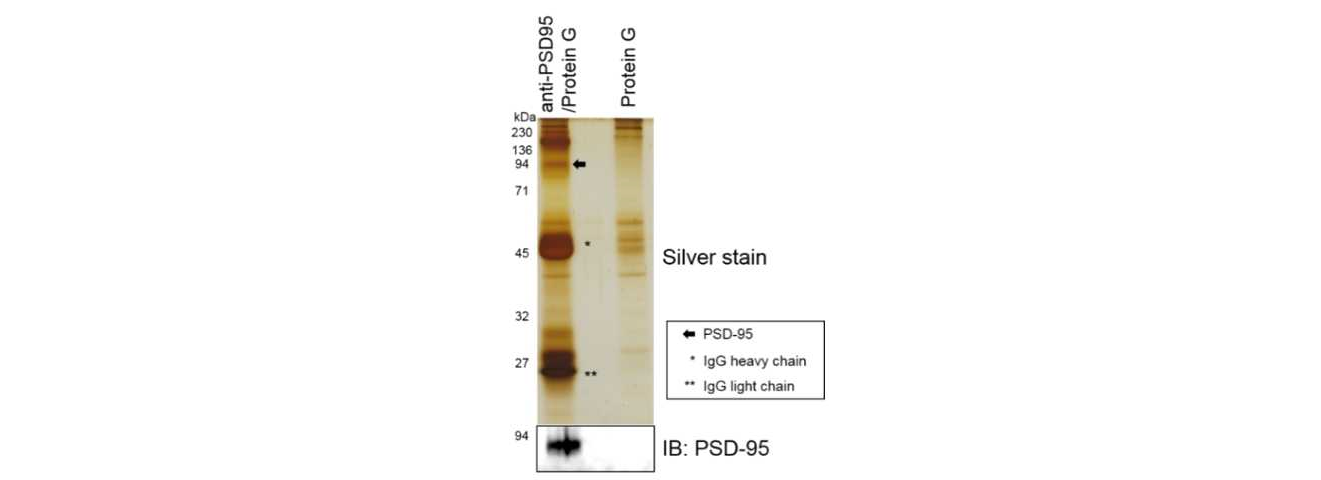
Solublized proteins with ULTRARIPA were subjected to immunoprecipitation. ULTRARIPA did not affect the immunoprecipitation.
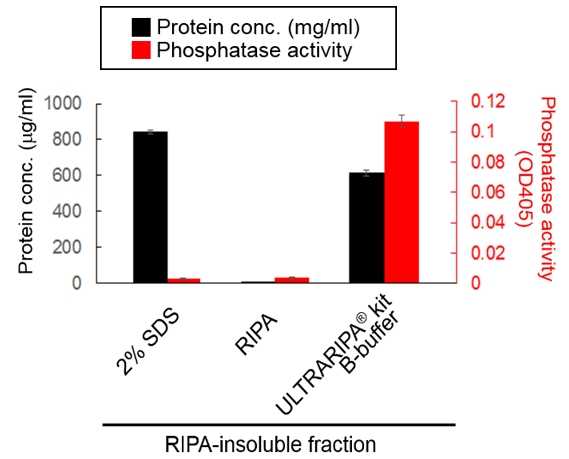
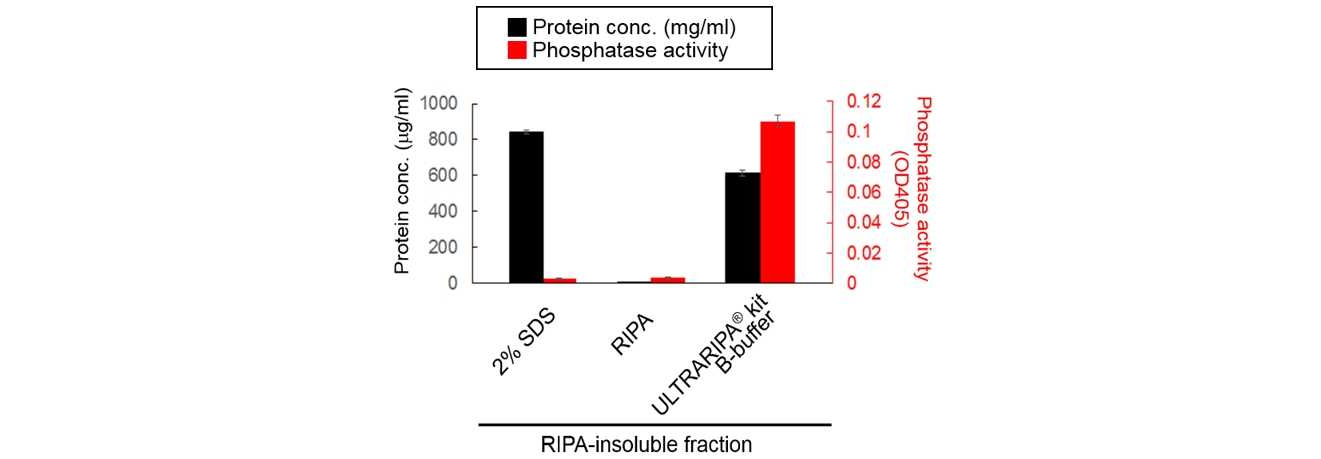
Phosphatase activity was maintained with ULTRARIPA extraction. On the other hand, The strong detergent denatured the PSD-95.
<Application 2>
Recovery of synaptic proteins
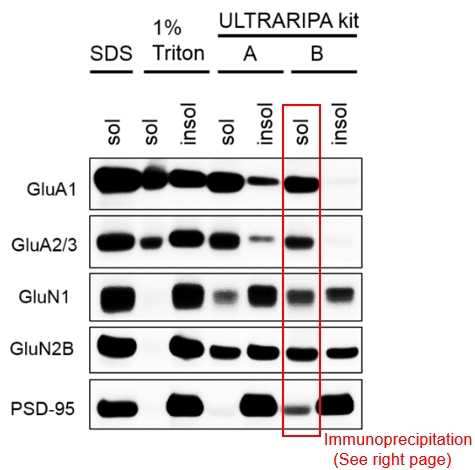
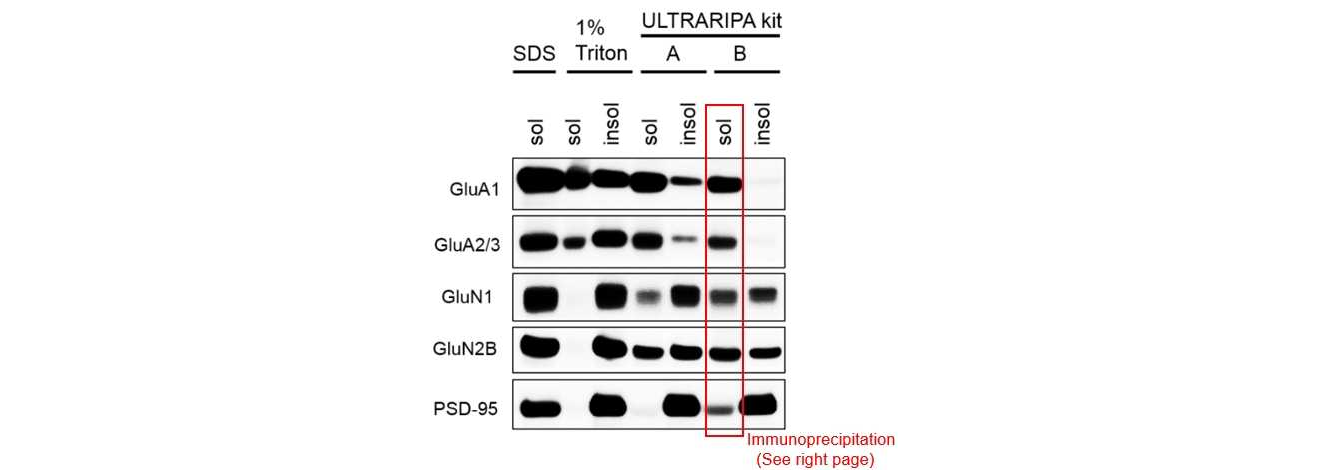
P2 membrane fraction of mouse brain tissue (hippocampus + cerebral cortex) was solubilized with each buffer. Then, insoluble fraction was solublized by adding 2%SDS. All proteins are solublized with ULTRARIPA. GluA1 and GluA2/3 were completely recovered.
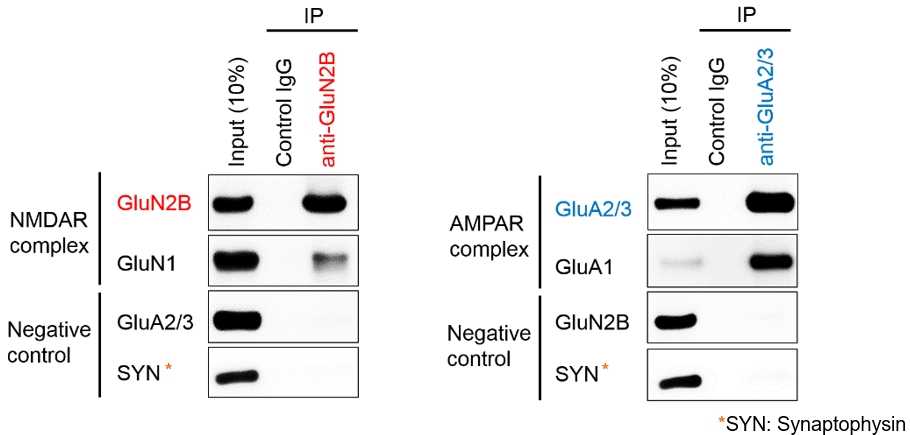
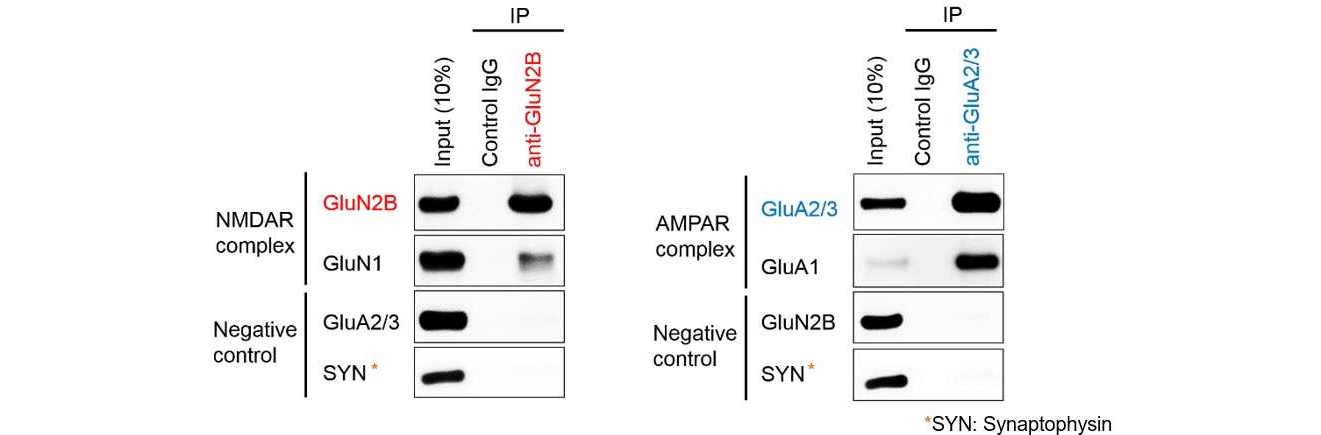
Solublized proteins with ULTRARIPA were subjected to immunoprecipitation and ULTRARIPA did not affect the immunoprecipitation. Physiological NMDAR and AMPAR complex is specifically detected by Immunoprecipitation. Best for analysis of protein complex in RIPA insoluble fraction.
<Application 3>
Recovery of synaptic proteins
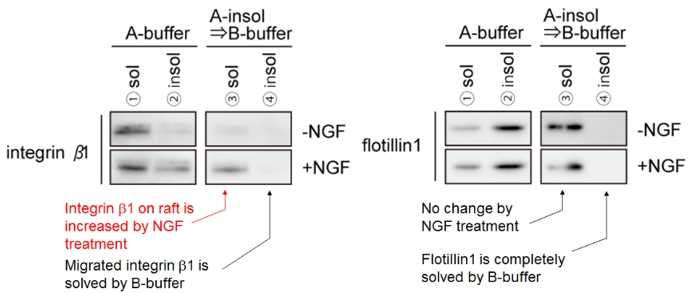
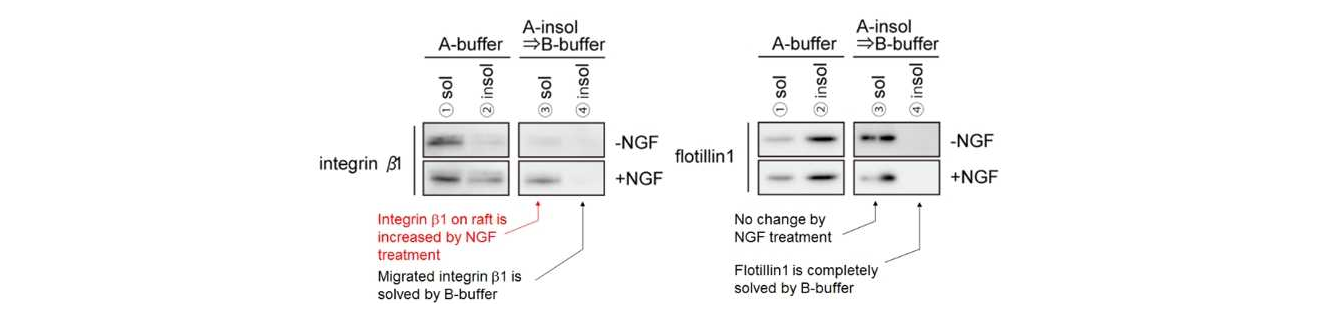
Mouse primary cultured DRG neurons were extracted with ULTRARIPA. Integrin β1 accumulated on lipid raft by NGF could be solublized with ULTRARIPA and Flotillin 1 was not changed by NGF stimulation. ULTRARIPA can be utilized for not only detecting the change of RIPA insoluble fraction dependent on external stimulus, but also detecting the change of protein complex dependent and detecting the change of enzyme activity in RIPA insoluble fraction on external stimulus.
BDL is a member of the Funakoshi Group.

